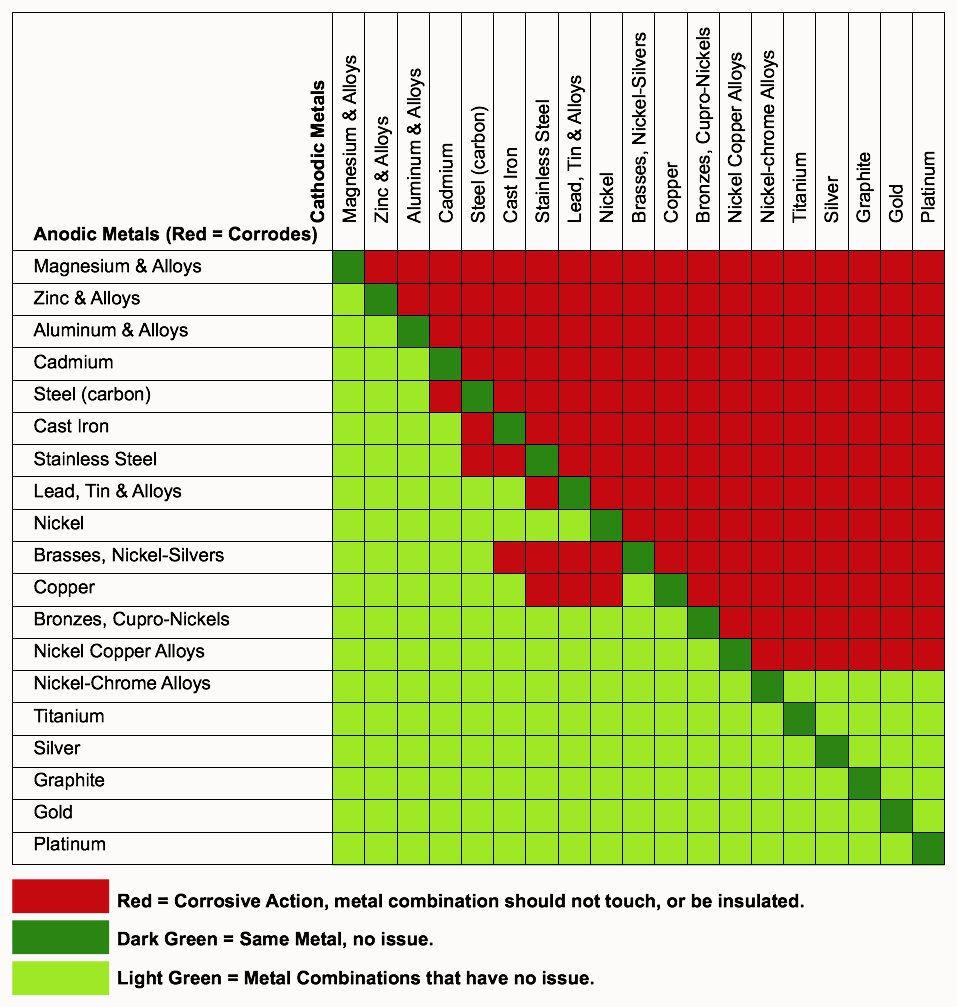
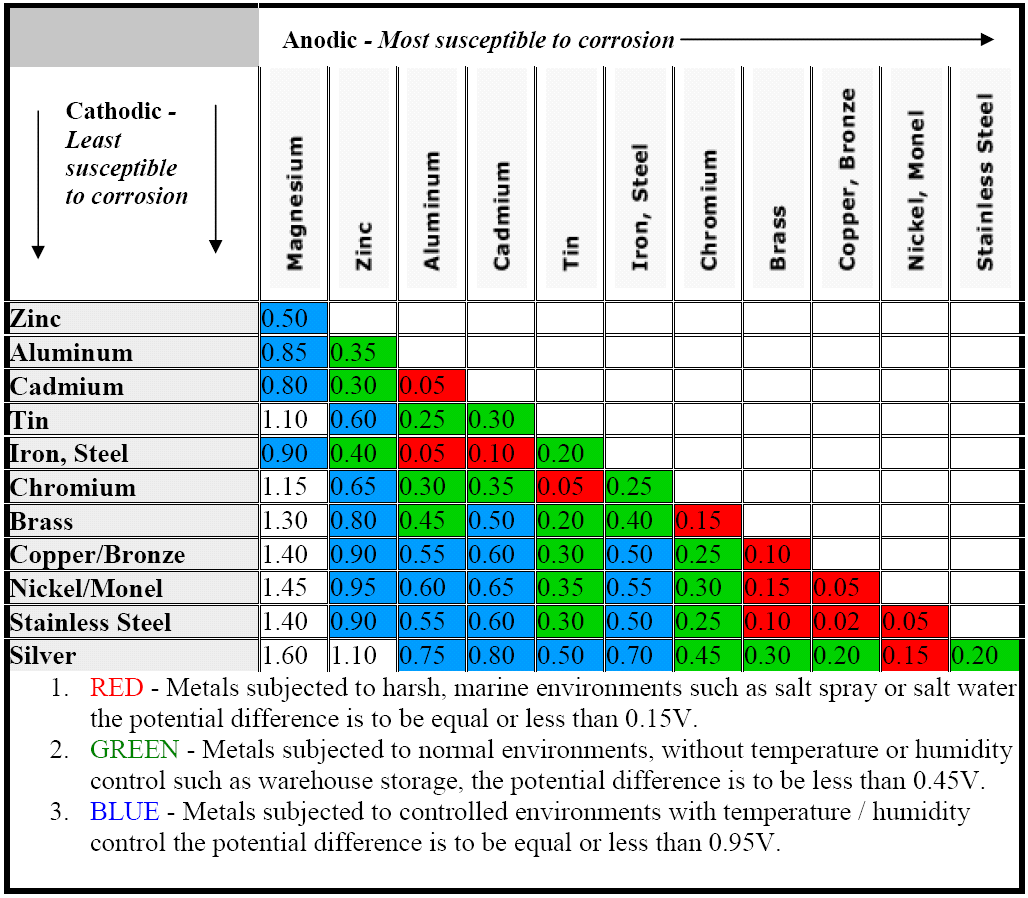
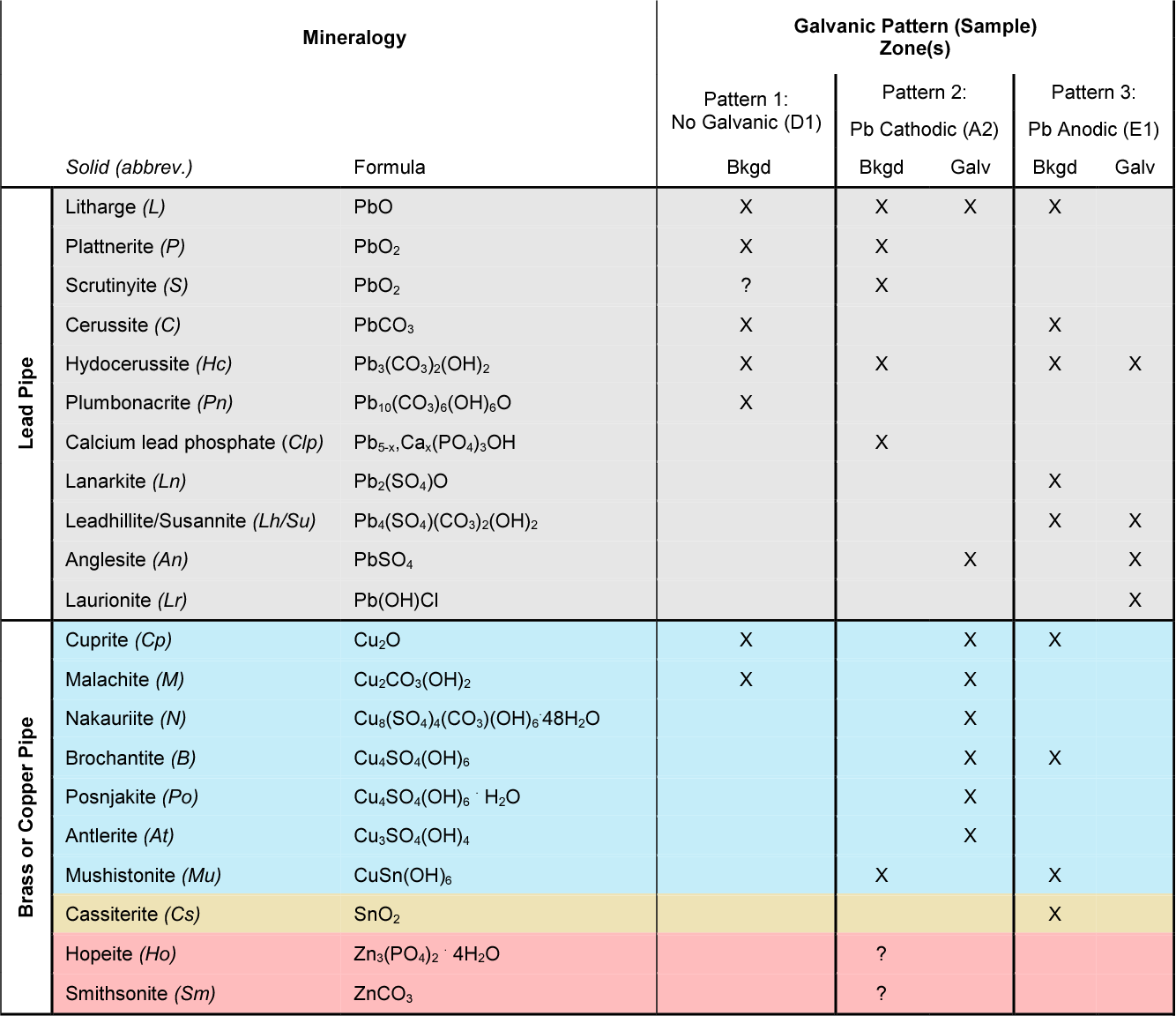
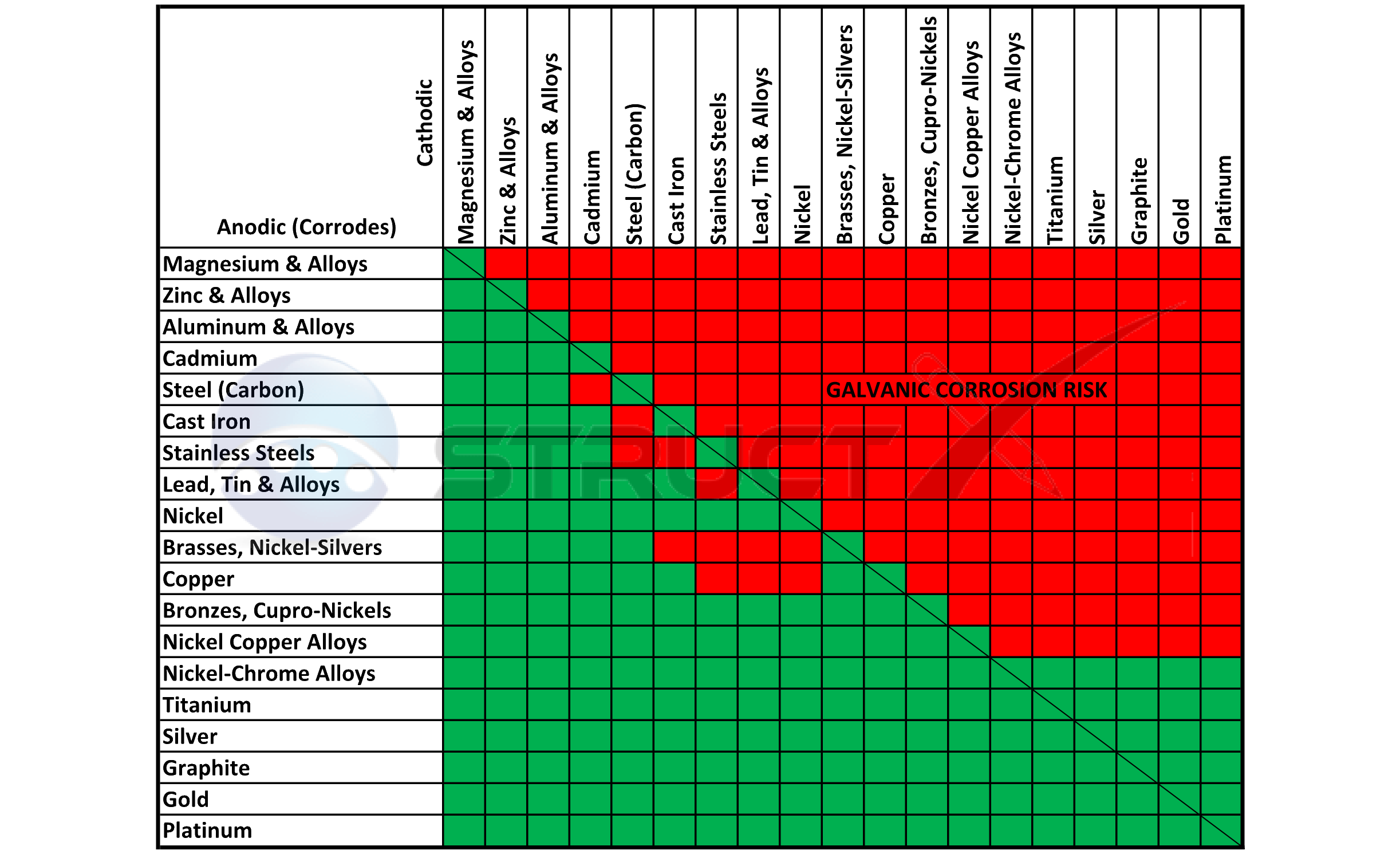
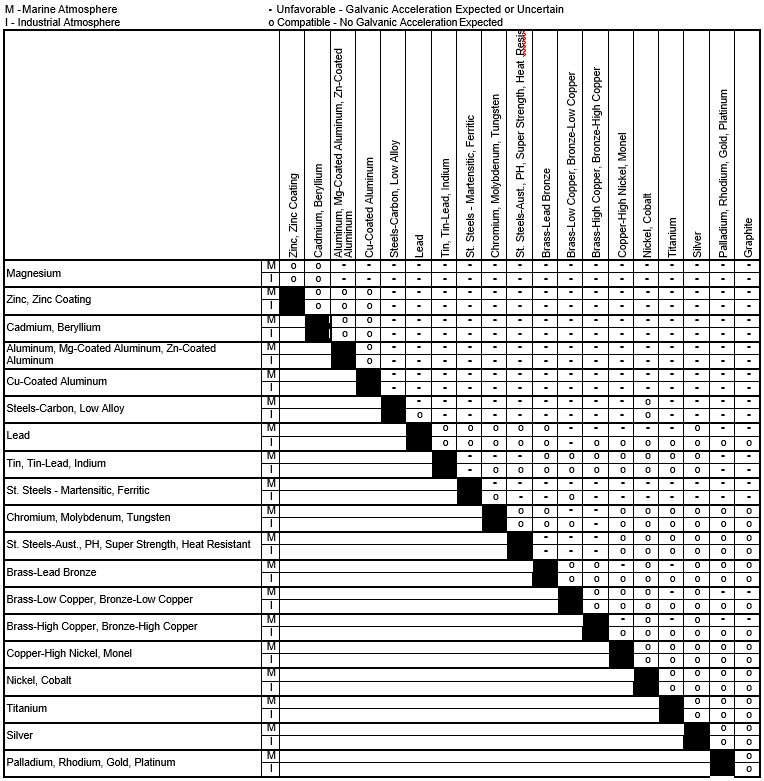
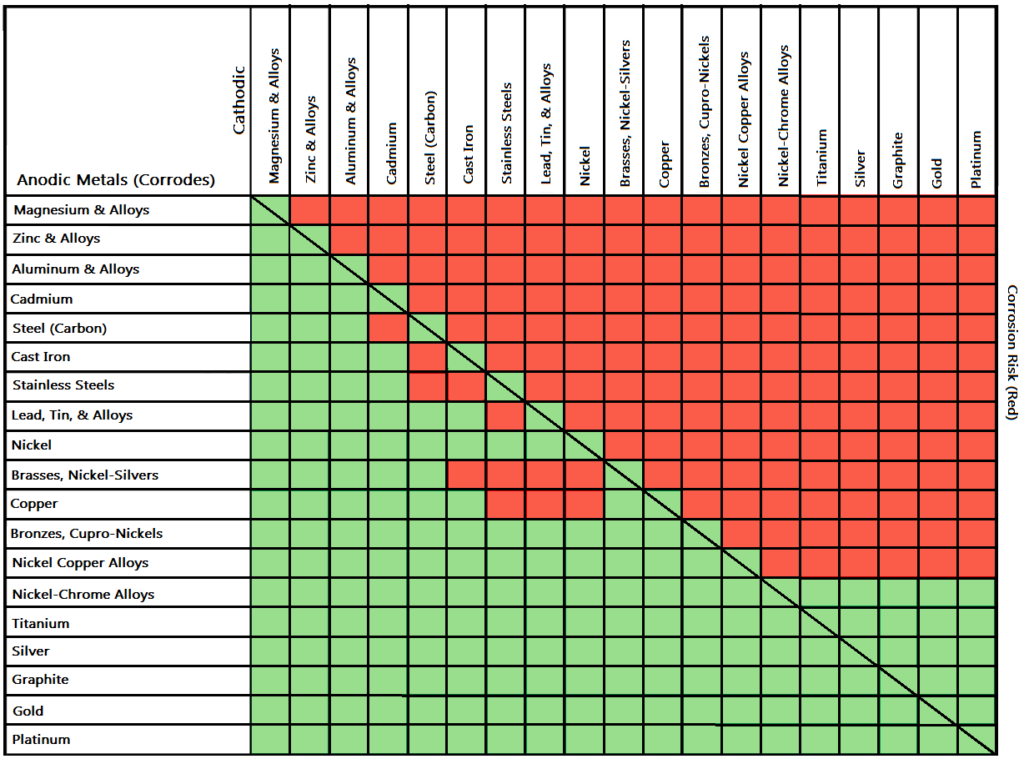
Dielectric Corrosion Chart
Dielectric Corrosion Chart - Web the following documents provide different points of view regarding the ranking of metals and coatings in practical schemes for preventing galvanic corrosion. Web this slide includes a chart of galvanic corrosion potential between common construction metals. For any combination of dissimilar metals, the metal with the lower number will act. Web galvanic corrosion is the damage of metal due to an electrochemical reaction between dissimilar metals in contact with an electrolyte. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. Web the following table was developed by interpreting available corrosion data and indicates the impact of electrical potential, environment, and surface area ratios to predict the. Web this article examines how dissimilar metals can lead to galvanic corrosion. Web below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal. The most active metals in the galvanic corrosion chart, like aluminum, zinc,. Web there are two primary types of galvanic cells that cause corrosion: The most active metals in the galvanic corrosion chart, like aluminum, zinc,. Web the following table was developed by interpreting available corrosion data and indicates the impact of electrical potential, environment, and surface area ratios to predict the. Web galvanic corrosion (also called ' dissimilar metal corrosion' or wrongly 'electrolysis') refers to corrosion damage induced when two dissimilar materials are coupled in a corrosive. Web metals listed on the top of the chart (anodic) will corrode faster than the metals on the bottom of the chart (cathodic). The alloys near the bottom are cathodic and. Web the galvanic corrosion table ranks metals from the most “active” to the least active. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. The corroded area was machined out and rebuilt with alloy 625 filler metal which is. Web the “galvanic series of metals and alloys” chart above provides a realistic and practical ranking of metallic electrical potentials. Second, there must be an. Web the following documents provide different points of view regarding the ranking of metals and coatings in practical schemes for preventing galvanic corrosion. See the chart with anodic, cathodic, and neutral. Contact a corrosion specialist to determine the best. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. Galvanic corrosion. Web a phenomenon known as galvanic corrosion occurs when dissimilar metals, subjected to the same environment, comprised of a conducting solution, are in direct electrical contact. Web find out how different metals will corrode when placed together in an assembly based on their galvanic corrosion potential. Web galvanic corrosion, also known as bimetallic corrosion or dissimilar metal corrosion, is an. The corroded area was machined out and rebuilt with alloy 625 filler metal which is. Web galvanic corrosion occurs when two different metals or alloys with different nobilities and therefore different electrochemical potentials come into contact with each. Web the galvanic corrosion table ranks metals from the most “active” to the least active. When dissimilar metals are used together in. Web galvanic corrosion, also known as bimetallic corrosion or dissimilar metal corrosion, is an electrochemical process that occurs when two different metals are in. Contact a corrosion specialist to determine the best. This phenomenon is named after italian ph… Galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when. Web this article examines how dissimilar metals can lead to galvanic corrosion. Second, there must be an. By eliminating any one of. For any combination of dissimilar metals, the metal with the lower number will act. Web galvanic corrosion occurs when two different metals or alloys with different nobilities and therefore different electrochemical potentials come into contact with each. Web this slide includes a chart of galvanic corrosion potential between common construction metals. Second, there must be an. It includes a chart that shows how different plating materials react to one another with. Web the following documents provide different points of view regarding the ranking of metals and coatings in practical schemes for preventing galvanic corrosion. Web galvanic corrosion. Web this article examines how dissimilar metals can lead to galvanic corrosion. Web the galvanic corrosion table ranks metals from the most “active” to the least active. The most active metals in the galvanic corrosion chart, like aluminum, zinc,. Web galvanic corrosion, also known as bimetallic corrosion or dissimilar metal corrosion, is an electrochemical process that occurs when two different. Web find out how different metals will corrode when placed together in an assembly based on their galvanic corrosion potential. Web metals listed on the top of the chart (anodic) will corrode faster than the metals on the bottom of the chart (cathodic). A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power. Web galvanic corrosion undermined the keeper rings, leading to failure and leakage. The most active metals in the galvanic corrosion chart, like aluminum, zinc,. Second, there must be an. Web the “galvanic series of metals and alloys” chart above provides a realistic and practical ranking of metallic electrical potentials. It includes a chart that shows how different plating materials react. Web galvanic corrosion undermined the keeper rings, leading to failure and leakage. Web this article examines how dissimilar metals can lead to galvanic corrosion. First there must be two electrochemically dissimilar metals present. For any combination of dissimilar metals, the metal with the lower number will act. Web this slide includes a chart of galvanic corrosion potential between common construction. Galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. Web metals listed on the top of the chart (anodic) will corrode faster than the metals on the bottom of the chart (cathodic). Web there are two primary types of galvanic cells that cause corrosion: Web the following table was developed by interpreting available corrosion data and indicates the impact of electrical potential, environment, and surface area ratios to predict the. Web the galvanic corrosion table ranks metals from the most “active” to the least active. Web this article examines how dissimilar metals can lead to galvanic corrosion. Web below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal. Web galvanic corrosion, also known as bimetallic corrosion or dissimilar metal corrosion, is an electrochemical process that occurs when two different metals are in. The corroded area was machined out and rebuilt with alloy 625 filler metal which is. The alloys near the bottom are cathodic and. See the chart with anodic, cathodic, and neutral. Web there are three conditions that must exist for galvanic corrosion to occur. Contact a corrosion specialist to determine the best. For any combination of dissimilar metals, the metal with the lower number will act. Web galvanic corrosion (also called ' dissimilar metal corrosion' or wrongly 'electrolysis') refers to corrosion damage induced when two dissimilar materials are coupled in a corrosive. Web find out how different metals will corrode when placed together in an assembly based on their galvanic corrosion potential.Galvanic Action Corrosion Prevention Architect's Blog
Dielectric Corrosion Chart A Visual Reference of Charts Chart Master
Dissimilar Corrosion Materials Tables
Dielectric Corrosion Chart A Visual Reference of Charts Chart Master
Galvanic Series (electrochemical series)
Corrosion charts Graphite Technology
Dielectric Corrosion Chart A Visual Reference of Charts Chart Master
Dielectric Chart
Galvanic Corrosion Common Questions Answered
Dielectric Corrosion Chart A Visual Reference of Charts Chart Master
The Most Active Metals In The Galvanic Corrosion Chart, Like Aluminum, Zinc,.
First There Must Be Two Electrochemically Dissimilar Metals Present.
Web Galvanic Corrosion Occurs When Two Different Metals Or Alloys With Different Nobilities And Therefore Different Electrochemical Potentials Come Into Contact With Each.
Web The “Galvanic Series Of Metals And Alloys” Chart Above Provides A Realistic And Practical Ranking Of Metallic Electrical Potentials.
Related Post: